|
Answers which suggest that the number of bonded ions depends upon the number of neighbours should be considered correct. 
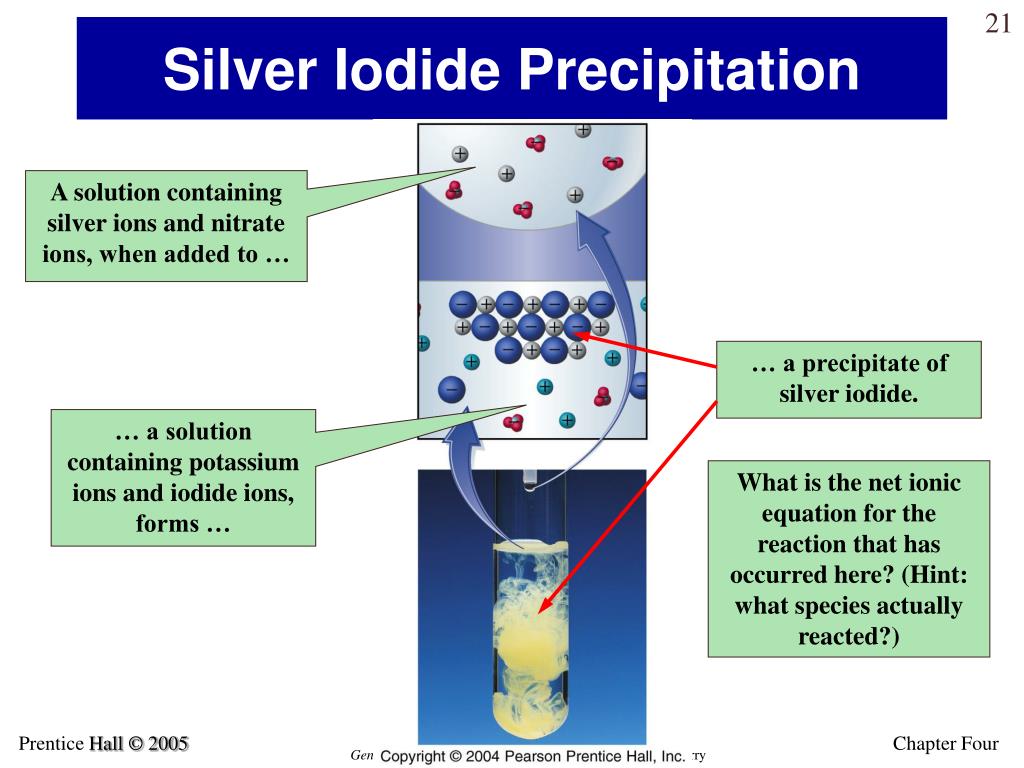
Students (especially those in the 14–16 age range) will not be expected to know about the precise crystal structure of silver chloride.(Answers about ion formation through electron transfer are wrong.) The important point is that the silver ions and chloride ions are already present in the mixture, and the reaction involves the electrical attraction causing the ions to clump together and form crystals.H 2O molecules, Na +, NO 3 –ions (also allow H +or H 3O +and OH –ions, as long as H 2O molecules given). NB unless care is taken with reacting quantities the final solution may also contain either silver ions or chloride ions - but not both.H 2O molecules, Ag +, NO 3 –ions (also allow H +or H 3O +and OH –ions, as long as H 2O molecules given).H 2O molecules, Na +, Cl –ions (also allow H +or H 3O +and OH –ions, as long as H 2O molecules given).How many iodide ions do you think might be bonded to each lead ion in lead iodide? (Give the reason for your answer, if you can.)Īnswers A reaction to form silver chloride.What do you think happens to the particles in the mixture, when the ionic bond forms in the lead iodide in this reaction?.What particles (such as particular atoms, molecules, ions) do you think are present in the liquid after it is filtered? The liquid that is left after filtration contains potassium nitrate. The solid can be separated from the liquid by filtration. The following reaction take place: potassium iodide(aq) + lead nitrate(aq) →potassium nitrate(aq) + lead iodide(s) The solid that is formed is lead iodide. When potassium iodide solution is mixed with lead nitrate solution a yellow solid forms.What particles (such as particular atoms, molecules, ions) do you think are present in lead nitrate solution? Lead nitrate dissolves in water to give lead nitrate solution.What particles (such as particular atoms, molecules, ions) do you think are present in potassium iodide solution? Potassium iodide dissolves in water to give potassium iodide solution.It can be prepared by reacting lead nitrate solution and potassium iodide solution. This type of chemical b_ is called ionic bonding. Each i_ is attracted to each of those counter ions surrounding it. Sodium ions (Na +) and chloride ions (Cl –) are bonded together by the e_ attraction between the positive and negative ions. When solutions of sodium chloride and silver nitrate are mixed, then a white solid (silver chloride) forms: sodium chloride(aq) + silver nitrate(aq) →sodium nitrate(aq) + silver chloride(s) Fill in the gaps below. This exercise is about what happens during a precipitation reaction. How many chloride ions do you think are bonded to each silver ion in silver chloride? (Give the reason for your answer, if you can.). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
